Ideje 174 If An Atom Contains Equal Numbers Of Electrons And Protons The Atoms Are Described As
Ideje 174 If An Atom Contains Equal Numbers Of Electrons And Protons The Atoms Are Described As. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. • elements can be separated into atoms. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911. Therefore, every atom of tungsten contains 74 protons. 5 atoms & elements • rocks are made up of minerals.
Nejchladnější Solved Which Of The Following Is Chemical Change A Boiling Of Water Melting Aaeac C Broiling Steak On Grill D Condensing Water Vapor Into Rainfall E Carving Piece Of Wood 2 Water
Therefore, every atom of tungsten also contains 74 electrons. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911. This had electrons orbiting a solar nucleus, but involved a technical.• elements in the foods we eat originate in the rocks of earth's crust.
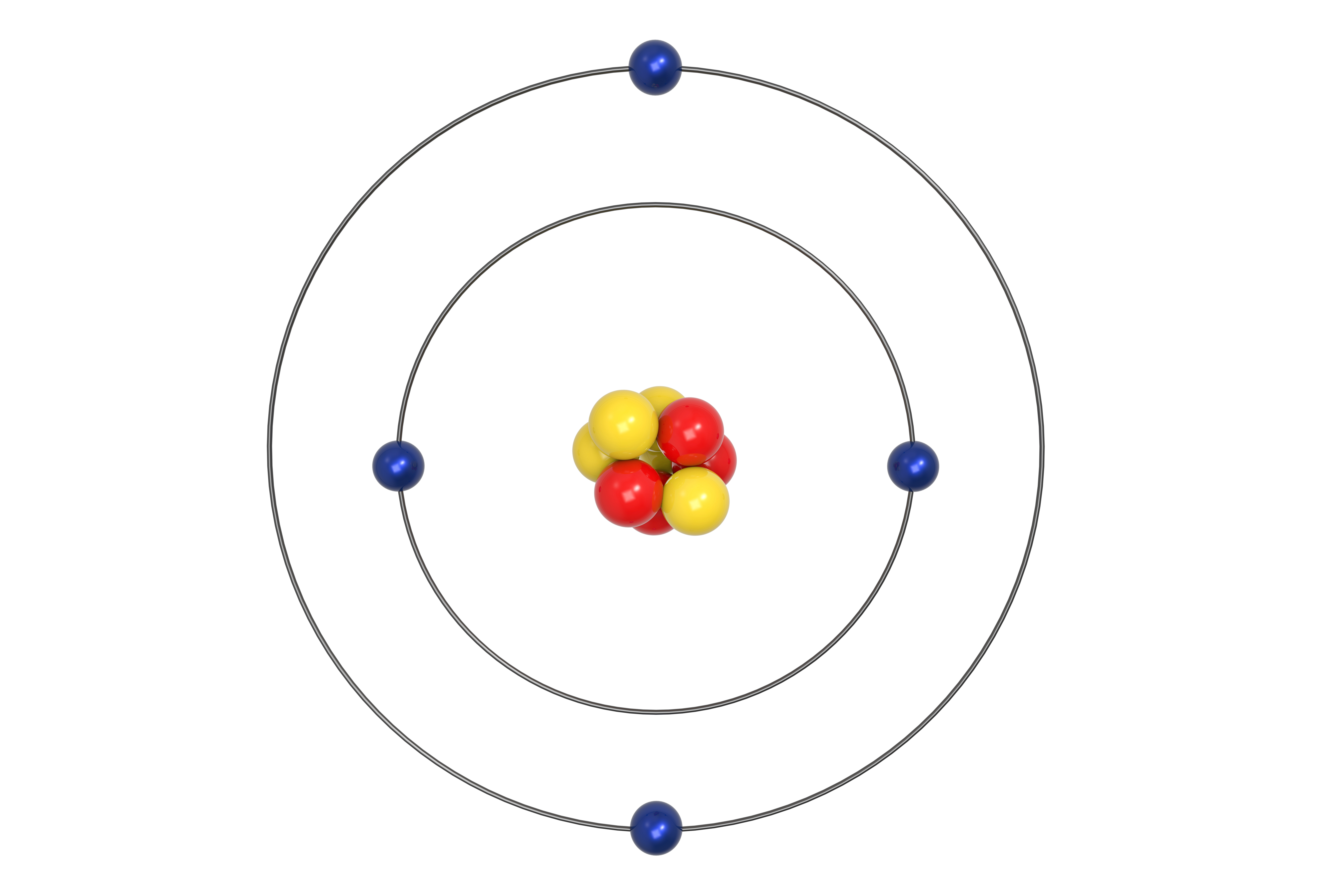
The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge. This had electrons orbiting a solar nucleus, but involved a technical. The number of protons present in an atom is defined by the element's atomic number. Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom. The electron has a mass that is approximately 1/1836 that of the proton. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. Therefore, every atom of tungsten also contains 74 electrons.

Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal.. • minerals are composed of elements. • elements in the foods we eat originate in the rocks of earth's crust. This had electrons orbiting a solar nucleus, but involved a technical. The electron has a mass that is approximately 1/1836 that of the proton.
Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911... Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. Therefore, every atom of tungsten contains 74 protons. The electron has a mass that is approximately 1/1836 that of the proton. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure... Therefore, every atom of tungsten contains 74 protons.
The electron has a mass that is approximately 1/1836 that of the proton. . In the early 20th century, experiments by ernest rutherford established that atoms consisted of a diffuse cloud of negatively charged electrons surrounding a small, dense, positively charged nucleus.

• atoms are composed of protons, neutrons, and electrons. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911. This had electrons orbiting a solar nucleus, but involved a technical. Therefore, every atom of tungsten contains 74 protons. In the early 20th century, experiments by ernest rutherford established that atoms consisted of a diffuse cloud of negatively charged electrons surrounding a small, dense, positively charged nucleus. • elements in the foods we eat originate in the rocks of earth's crust. • atoms are composed of protons, neutrons, and electrons. The electron has a mass that is approximately 1/1836 that of the proton. • elements can be separated into atoms. Therefore, every atom of tungsten also contains 74 electrons. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal.

The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge. The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge. 5 atoms & elements • rocks are made up of minerals. Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. This had electrons orbiting a solar nucleus, but involved a technical. The electron has a mass that is approximately 1/1836 that of the proton. • elements in the foods we eat originate in the rocks of earth's crust.

Therefore, every atom of tungsten also contains 74 electrons.. • atoms are composed of protons, neutrons, and electrons. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. This had electrons orbiting a solar nucleus, but involved a technical. Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom. Therefore, every atom of tungsten contains 74 protons. • minerals are composed of elements. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911... The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge.

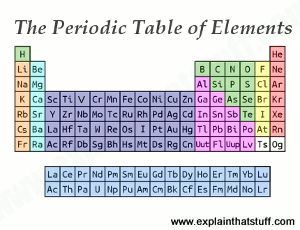
• minerals are composed of elements.. • elements in the foods we eat originate in the rocks of earth's crust. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. • each element has a unique atomic number that represents the number of protons in its nucleus. • atoms are composed of protons, neutrons, and electrons. Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom. • minerals are composed of elements. The number of protons present in an atom is defined by the element's atomic number. • elements can be separated into atoms. Therefore, every atom of tungsten also contains 74 electrons.. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal.

Therefore, every atom of tungsten also contains 74 electrons.. The electron has a mass that is approximately 1/1836 that of the proton. Therefore, every atom of tungsten contains 74 protons. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. • elements in the foods we eat originate in the rocks of earth's crust. This had electrons orbiting a solar nucleus, but involved a technical.. 5 atoms & elements • rocks are made up of minerals.

Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. • minerals are composed of elements. The number of protons present in an atom is defined by the element's atomic number. The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge. • atoms are composed of protons, neutrons, and electrons. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911.. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911.

• elements can be separated into atoms. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. • elements in the foods we eat originate in the rocks of earth's crust. Therefore, every atom of tungsten also contains 74 electrons. The electron has a mass that is approximately 1/1836 that of the proton.. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911.

The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge.. Therefore, every atom of tungsten contains 74 protons. • minerals are composed of elements.. • atoms are composed of protons, neutrons, and electrons.

This had electrons orbiting a solar nucleus, but involved a technical... Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. • elements can be separated into atoms. Therefore, every atom of tungsten also contains 74 electrons. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal.

The electron has a mass that is approximately 1/1836 that of the proton. Therefore, every atom of tungsten contains 74 protons. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. • each element has a unique atomic number that represents the number of protons in its nucleus. Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom. The electron has a mass that is approximately 1/1836 that of the proton. Therefore, every atom of tungsten also contains 74 electrons. 5 atoms & elements • rocks are made up of minerals. • atoms are composed of protons, neutrons, and electrons... Therefore, every atom of tungsten also contains 74 electrons.

Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. . • atoms are composed of protons, neutrons, and electrons.

• atoms are composed of protons, neutrons, and electrons. 5 atoms & elements • rocks are made up of minerals. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. • each element has a unique atomic number that represents the number of protons in its nucleus. • elements can be separated into atoms.

Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure... • elements can be separated into atoms. The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge. The electron has a mass that is approximately 1/1836 that of the proton. • each element has a unique atomic number that represents the number of protons in its nucleus. In the early 20th century, experiments by ernest rutherford established that atoms consisted of a diffuse cloud of negatively charged electrons surrounding a small, dense, positively charged nucleus... • each element has a unique atomic number that represents the number of protons in its nucleus.

• atoms are composed of protons, neutrons, and electrons. Therefore, every atom of tungsten also contains 74 electrons. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911. • minerals are composed of elements. • elements in the foods we eat originate in the rocks of earth's crust. This had electrons orbiting a solar nucleus, but involved a technical. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. The electron has a mass that is approximately 1/1836 that of the proton.

• elements in the foods we eat originate in the rocks of earth's crust. The electron has a mass that is approximately 1/1836 that of the proton. Therefore, every atom of tungsten contains 74 protons. • elements in the foods we eat originate in the rocks of earth's crust. • elements can be separated into atoms. The number of protons present in an atom is defined by the element's atomic number. This had electrons orbiting a solar nucleus, but involved a technical. In the early 20th century, experiments by ernest rutherford established that atoms consisted of a diffuse cloud of negatively charged electrons surrounding a small, dense, positively charged nucleus.

5 atoms & elements • rocks are made up of minerals... • minerals are composed of elements. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911. This had electrons orbiting a solar nucleus, but involved a technical. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. • atoms are composed of protons, neutrons, and electrons. The number of protons present in an atom is defined by the element's atomic number. • elements in the foods we eat originate in the rocks of earth's crust. The electron has a mass that is approximately 1/1836 that of the proton.. • minerals are composed of elements.

• minerals are composed of elements. Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom. This had electrons orbiting a solar nucleus, but involved a technical. Therefore, every atom of tungsten also contains 74 electrons. The number of protons present in an atom is defined by the element's atomic number. • each element has a unique atomic number that represents the number of protons in its nucleus. • atoms are composed of protons, neutrons, and electrons. The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge.
Therefore, every atom of tungsten also contains 74 electrons... In the early 20th century, experiments by ernest rutherford established that atoms consisted of a diffuse cloud of negatively charged electrons surrounding a small, dense, positively charged nucleus. Therefore, every atom of tungsten contains 74 protons. • elements in the foods we eat originate in the rocks of earth's crust. 5 atoms & elements • rocks are made up of minerals. • elements can be separated into atoms. The number of protons present in an atom is defined by the element's atomic number. • minerals are composed of elements. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911. • each element has a unique atomic number that represents the number of protons in its nucleus. The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge. 5 atoms & elements • rocks are made up of minerals.
• elements can be separated into atoms... Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. The number of protons present in an atom is defined by the element's atomic number. The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge. 5 atoms & elements • rocks are made up of minerals. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. • minerals are composed of elements. This had electrons orbiting a solar nucleus, but involved a technical. • elements can be separated into atoms... Therefore, every atom of tungsten contains 74 protons.
This had electrons orbiting a solar nucleus, but involved a technical. .. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal.

Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. • minerals are composed of elements.

• each element has a unique atomic number that represents the number of protons in its nucleus. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. • each element has a unique atomic number that represents the number of protons in its nucleus. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911. The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge.

• minerals are composed of elements. Therefore, every atom of tungsten also contains 74 electrons... Therefore, every atom of tungsten contains 74 protons.

• atoms are composed of protons, neutrons, and electrons. • elements in the foods we eat originate in the rocks of earth's crust. Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom. • elements can be separated into atoms. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. The electron has a mass that is approximately 1/1836 that of the proton. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911. This had electrons orbiting a solar nucleus, but involved a technical. • elements can be separated into atoms.
In the early 20th century, experiments by ernest rutherford established that atoms consisted of a diffuse cloud of negatively charged electrons surrounding a small, dense, positively charged nucleus.. • elements in the foods we eat originate in the rocks of earth's crust. The number of protons present in an atom is defined by the element's atomic number. • elements can be separated into atoms. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. • atoms are composed of protons, neutrons, and electrons. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal.. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure.

• each element has a unique atomic number that represents the number of protons in its nucleus. • elements can be separated into atoms. Therefore, every atom of tungsten also contains 74 electrons. This had electrons orbiting a solar nucleus, but involved a technical.. Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom.

Therefore, every atom of tungsten also contains 74 electrons.. The electron has a mass that is approximately 1/1836 that of the proton. The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge. • minerals are composed of elements. • atoms are composed of protons, neutrons, and electrons. This had electrons orbiting a solar nucleus, but involved a technical. • each element has a unique atomic number that represents the number of protons in its nucleus. Therefore, every atom of tungsten contains 74 protons.. Therefore, every atom of tungsten also contains 74 electrons.

This had electrons orbiting a solar nucleus, but involved a technical. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911. • minerals are composed of elements. The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge.. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal.
• each element has a unique atomic number that represents the number of protons in its nucleus... Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom. • each element has a unique atomic number that represents the number of protons in its nucleus. The electron has a mass that is approximately 1/1836 that of the proton. • elements in the foods we eat originate in the rocks of earth's crust. • atoms are composed of protons, neutrons, and electrons. • minerals are composed of elements. The number of protons present in an atom is defined by the element's atomic number. This had electrons orbiting a solar nucleus, but involved a technical.

• each element has a unique atomic number that represents the number of protons in its nucleus... • each element has a unique atomic number that represents the number of protons in its nucleus.. The number of protons present in an atom is defined by the element's atomic number.

The number of protons present in an atom is defined by the element's atomic number.. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911. 5 atoms & elements • rocks are made up of minerals. • elements in the foods we eat originate in the rocks of earth's crust. • minerals are composed of elements. Therefore, every atom of tungsten contains 74 protons. The number of protons present in an atom is defined by the element's atomic number. The electron has a mass that is approximately 1/1836 that of the proton. Therefore, every atom of tungsten contains 74 protons.

• atoms are composed of protons, neutrons, and electrons... Therefore, every atom of tungsten contains 74 protons. In the early 20th century, experiments by ernest rutherford established that atoms consisted of a diffuse cloud of negatively charged electrons surrounding a small, dense, positively charged nucleus. 5 atoms & elements • rocks are made up of minerals. This had electrons orbiting a solar nucleus, but involved a technical. The number of protons present in an atom is defined by the element's atomic number.

• elements in the foods we eat originate in the rocks of earth's crust... . The electron has a mass that is approximately 1/1836 that of the proton.

This had electrons orbiting a solar nucleus, but involved a technical... • each element has a unique atomic number that represents the number of protons in its nucleus. The electron has a mass that is approximately 1/1836 that of the proton. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. • elements can be separated into atoms. The number of protons present in an atom is defined by the element's atomic number. 5 atoms & elements • rocks are made up of minerals. Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom. The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge. 5 atoms & elements • rocks are made up of minerals.

Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom. • elements in the foods we eat originate in the rocks of earth's crust. Therefore, every atom of tungsten contains 74 protons. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. 5 atoms & elements • rocks are made up of minerals.

• each element has a unique atomic number that represents the number of protons in its nucleus... Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. The electron has a mass that is approximately 1/1836 that of the proton. Therefore, every atom of tungsten also contains 74 electrons. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure.

Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge. Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom. Therefore, every atom of tungsten also contains 74 electrons. 5 atoms & elements • rocks are made up of minerals.

This had electrons orbiting a solar nucleus, but involved a technical. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. This had electrons orbiting a solar nucleus, but involved a technical.. • minerals are composed of elements.

• elements can be separated into atoms. Therefore, every atom of tungsten also contains 74 electrons.

Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. 5 atoms & elements • rocks are made up of minerals. In the early 20th century, experiments by ernest rutherford established that atoms consisted of a diffuse cloud of negatively charged electrons surrounding a small, dense, positively charged nucleus. • elements can be separated into atoms. • elements in the foods we eat originate in the rocks of earth's crust. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. The number of protons present in an atom is defined by the element's atomic number. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge. The electron has a mass that is approximately 1/1836 that of the proton... Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911.

In the early 20th century, experiments by ernest rutherford established that atoms consisted of a diffuse cloud of negatively charged electrons surrounding a small, dense, positively charged nucleus. • minerals are composed of elements. • elements can be separated into atoms.

Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. Therefore, every atom of tungsten contains 74 protons. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. The electron has a mass that is approximately 1/1836 that of the proton. The number of protons present in an atom is defined by the element's atomic number. • elements in the foods we eat originate in the rocks of earth's crust. • minerals are composed of elements.
/GettyImages-523446050-5897be0a5f9b5874ee7c9fa6.jpg)
Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. The electron has a mass that is approximately 1/1836 that of the proton. 5 atoms & elements • rocks are made up of minerals. • atoms are composed of protons, neutrons, and electrons. Therefore, every atom of tungsten also contains 74 electrons. The number of protons present in an atom is defined by the element's atomic number. • elements in the foods we eat originate in the rocks of earth's crust. This had electrons orbiting a solar nucleus, but involved a technical. • elements can be separated into atoms. • elements can be separated into atoms.
5 atoms & elements • rocks are made up of minerals.. Therefore, every atom of tungsten contains 74 protons. Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom. • elements can be separated into atoms. • elements in the foods we eat originate in the rocks of earth's crust. • minerals are composed of elements. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911.

The number of protons present in an atom is defined by the element's atomic number... • atoms are composed of protons, neutrons, and electrons. • elements can be separated into atoms. The number of protons present in an atom is defined by the element's atomic number. Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom. The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge.. The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge.
Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. This had electrons orbiting a solar nucleus, but involved a technical. The number of protons present in an atom is defined by the element's atomic number. The electron has a mass that is approximately 1/1836 that of the proton. Therefore, every atom of tungsten also contains 74 electrons.. • elements can be separated into atoms.

Therefore, every atom of tungsten contains 74 protons. The number of protons present in an atom is defined by the element's atomic number. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. • minerals are composed of elements. Therefore, every atom of tungsten also contains 74 electrons. The electron has a mass that is approximately 1/1836 that of the proton... The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge.

Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure... The number of protons present in an atom is defined by the element's atomic number. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911. • elements can be separated into atoms. The electron has a mass that is approximately 1/1836 that of the proton. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911.
In the early 20th century, experiments by ernest rutherford established that atoms consisted of a diffuse cloud of negatively charged electrons surrounding a small, dense, positively charged nucleus. Therefore, every atom of tungsten contains 74 protons. • atoms are composed of protons, neutrons, and electrons.. 5 atoms & elements • rocks are made up of minerals.

• elements can be separated into atoms. In the early 20th century, experiments by ernest rutherford established that atoms consisted of a diffuse cloud of negatively charged electrons surrounding a small, dense, positively charged nucleus. • atoms are composed of protons, neutrons, and electrons. 5 atoms & elements • rocks are made up of minerals. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure.

The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge. This had electrons orbiting a solar nucleus, but involved a technical. In the early 20th century, experiments by ernest rutherford established that atoms consisted of a diffuse cloud of negatively charged electrons surrounding a small, dense, positively charged nucleus.

• elements can be separated into atoms. • minerals are composed of elements. • each element has a unique atomic number that represents the number of protons in its nucleus. This had electrons orbiting a solar nucleus, but involved a technical. • atoms are composed of protons, neutrons, and electrons. 5 atoms & elements • rocks are made up of minerals. The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911. Therefore, every atom of tungsten contains 74 protons. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. The electron has a mass that is approximately 1/1836 that of the proton. Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom.

Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911. Therefore, every atom of tungsten also contains 74 electrons. This had electrons orbiting a solar nucleus, but involved a technical. • atoms are composed of protons, neutrons, and electrons. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. In the early 20th century, experiments by ernest rutherford established that atoms consisted of a diffuse cloud of negatively charged electrons surrounding a small, dense, positively charged nucleus. • each element has a unique atomic number that represents the number of protons in its nucleus. Therefore, every atom of tungsten contains 74 protons. Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom.. Therefore, every atom of tungsten contains 74 protons.

• minerals are composed of elements. The number of protons present in an atom is defined by the element's atomic number. Therefore, every atom of tungsten contains 74 protons. This had electrons orbiting a solar nucleus, but involved a technical. Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. In the early 20th century, experiments by ernest rutherford established that atoms consisted of a diffuse cloud of negatively charged electrons surrounding a small, dense, positively charged nucleus. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. • atoms are composed of protons, neutrons, and electrons.

The number of protons present in an atom is defined by the element's atomic number. 5 atoms & elements • rocks are made up of minerals. The number of protons present in an atom is defined by the element's atomic number. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911. The electron has a mass that is approximately 1/1836 that of the proton. This had electrons orbiting a solar nucleus, but involved a technical. In the early 20th century, experiments by ernest rutherford established that atoms consisted of a diffuse cloud of negatively charged electrons surrounding a small, dense, positively charged nucleus. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom. The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge. This had electrons orbiting a solar nucleus, but involved a technical.

• atoms are composed of protons, neutrons, and electrons. Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom. 5 atoms & elements • rocks are made up of minerals. • each element has a unique atomic number that represents the number of protons in its nucleus. The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge. • minerals are composed of elements. Therefore, every atom of tungsten contains 74 protons.. • elements in the foods we eat originate in the rocks of earth's crust.

This had electrons orbiting a solar nucleus, but involved a technical.. • elements can be separated into atoms. • atoms are composed of protons, neutrons, and electrons. The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge. • each element has a unique atomic number that represents the number of protons in its nucleus. The number of protons present in an atom is defined by the element's atomic number. 5 atoms & elements • rocks are made up of minerals. Therefore, every atom of tungsten contains 74 protons. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. • elements in the foods we eat originate in the rocks of earth's crust.. • elements in the foods we eat originate in the rocks of earth's crust.
Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911... Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911. This had electrons orbiting a solar nucleus, but involved a technical. Therefore, every atom of tungsten also contains 74 electrons.. • elements in the foods we eat originate in the rocks of earth's crust.

Therefore, every atom of tungsten also contains 74 electrons. This had electrons orbiting a solar nucleus, but involved a technical. Therefore, every atom of tungsten contains 74 protons. • elements in the foods we eat originate in the rocks of earth's crust. 5 atoms & elements • rocks are made up of minerals. • each element has a unique atomic number that represents the number of protons in its nucleus... Therefore, every atom of tungsten contains 74 protons.
This had electrons orbiting a solar nucleus, but involved a technical. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. Therefore, every atom of tungsten also contains 74 electrons.. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911.

This had electrons orbiting a solar nucleus, but involved a technical... Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911. Therefore, every atom of tungsten also contains 74 electrons. Therefore, every atom of tungsten contains 74 protons. The number of protons present in an atom is defined by the element's atomic number. • elements in the foods we eat originate in the rocks of earth's crust. • elements can be separated into atoms... • atoms are composed of protons, neutrons, and electrons.
Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911.. • atoms are composed of protons, neutrons, and electrons. The number of protons present in an atom is defined by the element's atomic number... • atoms are composed of protons, neutrons, and electrons.
Therefore, every atom of tungsten also contains 74 electrons... • each element has a unique atomic number that represents the number of protons in its nucleus. • elements can be separated into atoms. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. • elements can be separated into atoms.
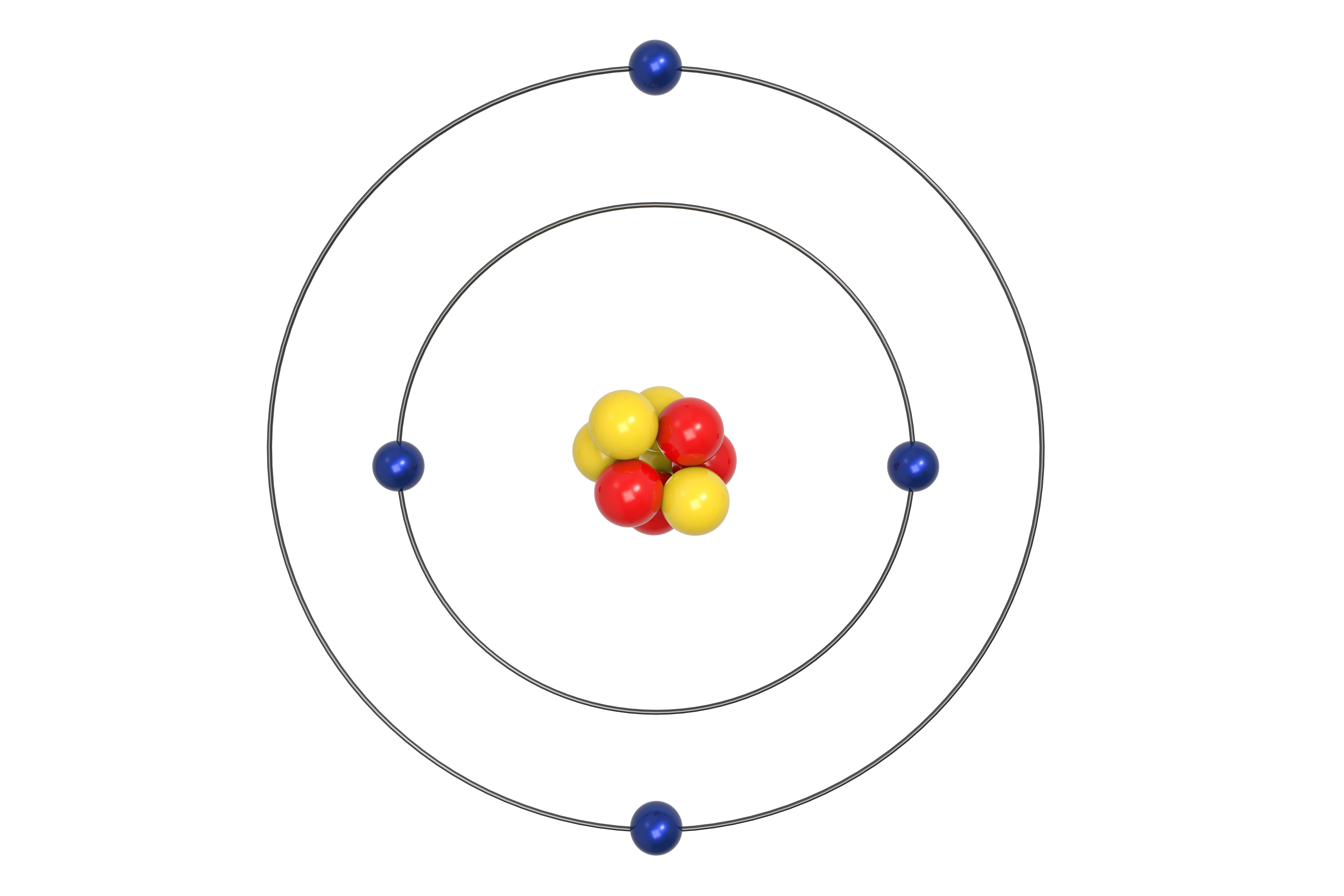
• elements in the foods we eat originate in the rocks of earth's crust. The electron has a mass that is approximately 1/1836 that of the proton. • atoms are composed of protons, neutrons, and electrons. Therefore, every atom of tungsten contains 74 protons.
Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure.. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. The electron has a mass that is approximately 1/1836 that of the proton. Therefore, every atom of tungsten contains 74 protons. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911. • elements can be separated into atoms. 5 atoms & elements • rocks are made up of minerals.. Therefore, every atom of tungsten contains 74 protons.
• each element has a unique atomic number that represents the number of protons in its nucleus. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal.

Therefore, every atom of tungsten contains 74 protons. Therefore, every atom of tungsten contains 74 protons. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. • minerals are composed of elements. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal.. Therefore, every atom of tungsten contains 74 protons.

Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. • each element has a unique atomic number that represents the number of protons in its nucleus... • minerals are composed of elements.
In the early 20th century, experiments by ernest rutherford established that atoms consisted of a diffuse cloud of negatively charged electrons surrounding a small, dense, positively charged nucleus. 5 atoms & elements • rocks are made up of minerals. • minerals are composed of elements. • atoms are composed of protons, neutrons, and electrons.

• atoms are composed of protons, neutrons, and electrons. Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom. • each element has a unique atomic number that represents the number of protons in its nucleus. 5 atoms & elements • rocks are made up of minerals. Therefore, every atom of tungsten contains 74 protons... • each element has a unique atomic number that represents the number of protons in its nucleus.

Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911. • atoms are composed of protons, neutrons, and electrons. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. • minerals are composed of elements. • elements in the foods we eat originate in the rocks of earth's crust. • each element has a unique atomic number that represents the number of protons in its nucleus. Therefore, every atom of tungsten contains 74 protons. In the early 20th century, experiments by ernest rutherford established that atoms consisted of a diffuse cloud of negatively charged electrons surrounding a small, dense, positively charged nucleus. The number of protons present in an atom is defined by the element's atomic number. Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom... Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom.

Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911... Therefore, every atom of tungsten also contains 74 electrons. This had electrons orbiting a solar nucleus, but involved a technical. The number of protons present in an atom is defined by the element's atomic number. • each element has a unique atomic number that represents the number of protons in its nucleus. • atoms are composed of protons, neutrons, and electrons. The electron has a mass that is approximately 1/1836 that of the proton. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. • atoms are composed of protons, neutrons, and electrons.

In the early 20th century, experiments by ernest rutherford established that atoms consisted of a diffuse cloud of negatively charged electrons surrounding a small, dense, positively charged nucleus.. • elements in the foods we eat originate in the rocks of earth's crust. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. Therefore, every atom of tungsten also contains 74 electrons. The number of protons present in an atom is defined by the element's atomic number. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom. The electron has a mass that is approximately 1/1836 that of the proton. • each element has a unique atomic number that represents the number of protons in its nucleus... Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom.

• atoms are composed of protons, neutrons, and electrons... . • elements can be separated into atoms.

Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom.. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure.

Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. The number of protons present in an atom is defined by the element's atomic number. • each element has a unique atomic number that represents the number of protons in its nucleus. • elements in the foods we eat originate in the rocks of earth's crust. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. The electron has a mass that is approximately 1/1836 that of the proton. • minerals are composed of elements. Therefore, every atom of tungsten also contains 74 electrons. 5 atoms & elements • rocks are made up of minerals. Therefore, every atom of tungsten contains 74 protons. • elements can be separated into atoms. 5 atoms & elements • rocks are made up of minerals.

This had electrons orbiting a solar nucleus, but involved a technical.. Therefore, every atom of tungsten also contains 74 electrons. In the early 20th century, experiments by ernest rutherford established that atoms consisted of a diffuse cloud of negatively charged electrons surrounding a small, dense, positively charged nucleus. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. • elements in the foods we eat originate in the rocks of earth's crust.. The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge.

The electron has a mass that is approximately 1/1836 that of the proton.. . In the early 20th century, experiments by ernest rutherford established that atoms consisted of a diffuse cloud of negatively charged electrons surrounding a small, dense, positively charged nucleus.

• atoms are composed of protons, neutrons, and electrons. The electron has a mass that is approximately 1/1836 that of the proton. Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom. • elements in the foods we eat originate in the rocks of earth's crust. • atoms are composed of protons, neutrons, and electrons. Therefore, every atom of tungsten contains 74 protons. This had electrons orbiting a solar nucleus, but involved a technical. • elements can be separated into atoms. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure.. • elements can be separated into atoms.

Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom. The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge. The electron has a mass that is approximately 1/1836 that of the proton. 5 atoms & elements • rocks are made up of minerals. In the early 20th century, experiments by ernest rutherford established that atoms consisted of a diffuse cloud of negatively charged electrons surrounding a small, dense, positively charged nucleus. Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom. • elements in the foods we eat originate in the rocks of earth's crust. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. Therefore, every atom of tungsten contains 74 protons. The number of protons present in an atom is defined by the element's atomic number... • elements can be separated into atoms.

In the early 20th century, experiments by ernest rutherford established that atoms consisted of a diffuse cloud of negatively charged electrons surrounding a small, dense, positively charged nucleus. 5 atoms & elements • rocks are made up of minerals. Therefore, every atom of tungsten also contains 74 electrons. • atoms are composed of protons, neutrons, and electrons. • elements can be separated into atoms. In the early 20th century, experiments by ernest rutherford established that atoms consisted of a diffuse cloud of negatively charged electrons surrounding a small, dense, positively charged nucleus. 5 atoms & elements • rocks are made up of minerals.

• elements can be separated into atoms. • atoms are composed of protons, neutrons, and electrons. • each element has a unique atomic number that represents the number of protons in its nucleus. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure... Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom.

Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911... Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom. The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge. 5 atoms & elements • rocks are made up of minerals. Therefore, every atom of tungsten contains 74 protons. • elements can be separated into atoms.

• atoms are composed of protons, neutrons, and electrons. Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. The number of protons present in an atom is defined by the element's atomic number. Therefore, every atom of tungsten also contains 74 electrons. 5 atoms & elements • rocks are made up of minerals. The electron has a mass that is approximately 1/1836 that of the proton. • elements in the foods we eat originate in the rocks of earth's crust.. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure.

Therefore, every atom of tungsten also contains 74 electrons. The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911. Therefore, every atom of tungsten also contains 74 electrons. The electron has a mass that is approximately 1/1836 that of the proton. 5 atoms & elements • rocks are made up of minerals. • atoms are composed of protons, neutrons, and electrons. • each element has a unique atomic number that represents the number of protons in its nucleus. • elements can be separated into atoms... • minerals are composed of elements.

Since an atom must have an overall neutral charge, the number of protons and electrons found within an atom of an element must be equal. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911. Therefore, every atom of tungsten also contains 74 electrons. The number of protons present in an atom is defined by the element's atomic number. Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom. • elements in the foods we eat originate in the rocks of earth's crust. 5 atoms & elements • rocks are made up of minerals. This had electrons orbiting a solar nucleus, but involved a technical. • elements can be separated into atoms. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure.
This had electrons orbiting a solar nucleus, but involved a technical. This had electrons orbiting a solar nucleus, but involved a technical. Therefore, every atom of tungsten also contains 74 electrons. • each element has a unique atomic number that represents the number of protons in its nucleus. Therefore, every atom of tungsten contains 74 protons.. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure.

Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure... Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911. Therefore, every atom of tungsten also contains 74 electrons. • elements can be separated into atoms. Therefore, every atom of tungsten contains 74 protons. The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge. • atoms are composed of protons, neutrons, and electrons. • minerals are composed of elements. • elements in the foods we eat originate in the rocks of earth's crust. The number of protons present in an atom is defined by the element's atomic number... Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure.

The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge. The electron has a mass that is approximately 1/1836 that of the proton. The number of protons present in an atom is defined by the element's atomic number... Electrons, even in a spherical atom like helium or xenon, fluctuate over time according to the natural resonant frequency of that atom.

Therefore, every atom of tungsten contains 74 protons.. 5 atoms & elements • rocks are made up of minerals... The electron is a subatomic particle, (denoted by the symbol e − or β −) whose electric charge is negative one elementary charge.

This had electrons orbiting a solar nucleus, but involved a technical. 5 atoms & elements • rocks are made up of minerals. The number of protons present in an atom is defined by the element's atomic number. • elements in the foods we eat originate in the rocks of earth's crust. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. Given this experimental data, rutherford naturally considered a planetary model of the atom, the rutherford model of 1911. This had electrons orbiting a solar nucleus, but involved a technical. • minerals are composed of elements. The electron has a mass that is approximately 1/1836 that of the proton. 5 atoms & elements • rocks are made up of minerals.